So2cl2 Molecular Geometry
In the SO2Cl2 lewis structure, the whole electron density lies around the central S atom, and two Cl and two O are present at four sites of the tetrahedral moiety. The ideal bond angle should be 109.5 0 for tetrahedral but here the scenario is different.

Draw the Lewis structure for SO2Cl2 and then answer questions that
Sulfuryl chloride (SO 2 Cl 2) contains one sulfur atom, two chlorine atoms and two oxygen atoms. In SO 2 Cl 2 lewis structure, there are two double bonds between sulfur atom and oxygen atoms. Between sulfur and chlorine atoms, there are single bonds. No lone pairs exist on sulfur atom. Lewis structure of SO 2 Cl 2

SO2 Lewis Structure ,Valence Electrons ,Formal Charge Lewis Structure
A step-by-step explanation of how to draw the SOCl2 Lewis Dot Structure (Thionyl chloride).For the SOCl2 structure use the periodic table to find the total n.
A quick explanation of the molecular geometry of SOCl2 including a description of the SOCl2 bond angles.Looking at the SOCl2 Lewis structure we can see that.
[Solved] draw lewis structure and molecular geometry of xeo4 SO2CL2
It is possible to draw a structure with a double bond between a boron atom and a fluorine atom in BF 3, satisfying the octet rule, but experimental evidence indicates the bond lengths are closer to that expected for B-F single bonds. This suggests the best Lewis structure has three B-F single bonds and an electron deficient boron.
SO2Cl2 (Sulfuryl chloride) Lewis Structure
The Lewis structure for SOCl 2 requires you to place more than 8 valence electrons on Sulfur (S). You might think you've got the correct Lewis structure for SOCl 2 at first. Remember, Sulfur is in Period 3 and can hold more than 8 valence electrons. You'll want to calculate the formal charges on each atom to make sure you have the best Lewis.

Lewis dot structure for SO2Cl2 Sulfuryl chloride YouTube
A step-by-step explanation of how to draw the SO2Cl2 Lewis Dot Structure.For the SO2Cl2 structure use the periodic table to find the total number of valence.

SOCl2 Lewis Structure Thionyl chloride YouTube
Structure Sulfur is tetrahedral in SO 2 Cl 2 and the oxidation state of the sulfur atom is +6, as in sulfuric acid . Synthesis SO 2 Cl 2 is prepared by the reaction of sulfur dioxide and chlorine in the presence of a catalyst, such as activated carbon. [2] SO 2 + Cl 2 → SO 2 Cl 2 The product can be purified by fractional distillation .

H2S Lewis StructureThis post is about the lewis structure of hydrogen
To find formal charges in a Lewis structure, for each atom, you should count how many electrons it "owns". Count all of its lone pair electrons, and half of its bonding electrons. The difference between the atom's number of valence electrons and the number it owns is the formal charge. For example, in NH 3, N has 1 lone pair (2 electrons) and 3.

what is the lewis dot structure of SO2Cl2 ? Brainly.in
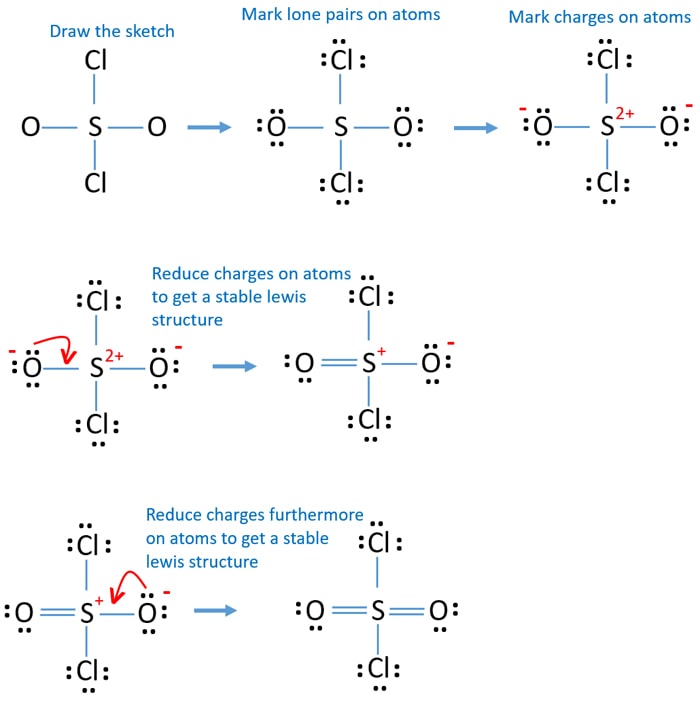
In the SO 2 Cl 2 Lewis structure, there are two single bonds and two double bonds around the sulfur atom, with two chlorine atoms and two oxygen atoms attached to it. Each chlorine atom has three lone pairs, and each oxygen atom has two lone pairs. Lewis dot structure for SO2Cl2 Sulfuryl chloride Watch on Contents Steps #1 First draw a rough sketch

So2Cl2 Lewis Structure
6 Steps to Draw the Lewis Structure of SOCl2 Step #1: Calculate the total number of valence electrons Here, the given molecule is SOCl2. In order to draw the lewis structure of SOCl2, first of all you have to find the total number of valence electrons present in the SOCl2 molecule.
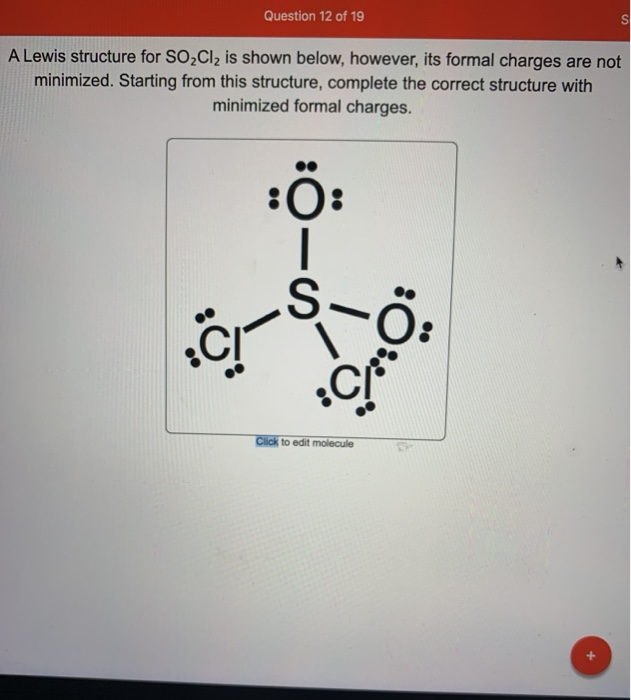
[Solved] Having trouble with a lewis structure for SO2Cl2 (s central
Step 1: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. When drawing the structure of an ion, be sure to add/subtract electrons to account for the charge. Step 2: Connect the atoms to each other with single bonds to form a "skeleton structure.".

SO2Cl2 Lewis Structure (Sulfuryl Chloride) YouTube
1 2 3 4 5 6 7 8 9 Share 1.7K views 1 year ago Lewis Structure SO2Cl2 is a chemical formula for Sulfuryl chloride. It is an inorganic compound, and to know more about its properties; we shall.

draw the lewis structure of SOCl2 Brainly.in
In socl2 lewis structure we see that as S has larger size and also less electronegative than other atoms it works as central atom in socl2. Sulphur has 6 electrons in the outermost shell out of which 4 electrons participate in sigma bonding with 2 cl atoms and 1 O atom. With O atom S forms 1 sigma and 1 Pi bond.
[Solved] Having trouble with a lewis structure for SO2Cl2 (s central
Lewis structure of SO2Cl2 contains double bonds between the Sulfur (S) atom & Oxygen (O) atoms and single bonds between the Sulfur (S) atom and Chlorine (Cl) atoms. The Sulfur atom (S) is at the center and it is surrounded by 2 Oxygen atoms (O) and 2 Chlorine atoms (Cl). Let's draw and understand this lewis dot structure step by step.
More Lewis Structures Worksheet Answers Ivuyteq
Chemistry Chemistry questions and answers Draw the Lewis Structure for SO2Cl2 and then answer questions that follow. -Do not include overall ion charges or formal charges in your drawing. -Do not include double bonds to oxygen atoms unless they are needed for the central atom to obey to octet rule. Questions: 1).